
Conjugated polymers have attracted significant attention as active materials in organic electronics, including organic photovoltaics, light-emitting diodes and thin-film transistors. Their appeal lies in the combination of solution processability, tunable electronic properties and mechanical flexibility, which together enable applications not readily achievable with conventional inorganic semiconductors. The widespread adoption of these materials, however, is hindered by their limited long-term stability. Exposure to oxygen, moisture, light and thermal stress can initiate a variety of degradation pathways, leading to structural and electronic changes that compromise device performance. Understanding the molecular level processes underlying such degradation remains a critical challenge for advancing the durability of organic electronic technologies.
Spectroscopic techniques are essential for probing the electronic structure and chemical evolution of conjugated polymers under operational and accelerated aging conditions. Such conventional methods as ultraviolet photoelectron spectroscopy (UPS) and X-ray photoelectron spectroscopy (XPS) reveal energy level alignment and chemical bonding, while device-based techniques like deep-level transient spectroscopy (DLTS), impedance spectroscopy and thermally stimulated current (TSC) measurements are used to identify trap or defect states. These approaches, however, often rely on device fabrication and can suffer from interface or contact artifacts.
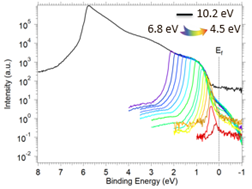
In contrast, Constant Final State Yield Spectroscopy (CFSYS) and Variable Energy Ultraviolet Photoelectron Spectroscopy (VE-UPS) offer a highly sensitive, device-free framework for mapping the occupied electronic structure of materials. CFSYS monitors electron emission at a fixed electron kinetic energy while sweeping the photon energy, providing energy references directly tied to the vacuum level and revealing defect state distributions intrinsic to the material. In contrast, VE-UPS varies the photon energy to obtain full UPS spectra at each excitation energy, offering detailed insight into the electronic structure beyond simple HOMO–LUMO transitions. Together, CFSYS and VE-UPS enhance sensitivity to subtle electronic variations and defect states that arise during degradation.
Applying these complementary techniques to conjugated polymer degradation enables a direct correlation between spectroscopic signatures and performance loss, providing new insight into the molecular origins of instability and guiding the design of more durable conjugated systems for organic electronic applications.
The work encompasses the development, optimization and application of advanced photoemission spectroscopy techniques to probe the electronic structure and stability of organic and hybrid materials. The second chapter focuses on precise calibration and performance optimization of the PHOIBOS hemispherical analyzer, ensuring accurate and reproducible spectroscopic measurements across diverse material systems. The third chapter highlights the design, construction and implementation of the VE-UPS and CFSYS systems, developed to enhance sensitivity to defect states and quantify the change in defect states of doped systems.
These techniques are applied in the fourth chapter to investigate defect evolution and stability in conjugated polymers under varying electrochemical doping levels, revealing how doping influences degradation and electronic structure. The final chapter extends these methods to Dion-Jacobson tin halide perovskites, demonstrating how variations in spacer cations modulate their structural, energetic, and optical properties. Together, these studies advance both the methodological capabilities and the fundamental understanding of material electronic behavior and stability.
 Abstract: Organic semiconductors, derived from π-conjugated polymers and molecules, enable the development of deformable, stretchable and flexible electronics due to their tunable redox, optical, electronic and mechanical properties. However, an informed understanding of how multi-scale morphological characteristics of the polymeric and molecular semiconductors influence bulk properties that contribute to electronic and optical performance, especially under operational thermal and mechanical stresses, remains incomplete.
Abstract: Organic semiconductors, derived from π-conjugated polymers and molecules, enable the development of deformable, stretchable and flexible electronics due to their tunable redox, optical, electronic and mechanical properties. However, an informed understanding of how multi-scale morphological characteristics of the polymeric and molecular semiconductors influence bulk properties that contribute to electronic and optical performance, especially under operational thermal and mechanical stresses, remains incomplete. 


 Abstract:
Abstract:  Noncovalent interactions (NCIs) in π-conjugated organic materials serve as tunable levers that influence molecular structure and intermolecular interactions in the condensed phase and, in turn, impact the electronic, optical and mechanical properties of these materials. NCIs include attractive dispersion, electrostatic and induction interactions as well as repulsive exchange interactions.
Noncovalent interactions (NCIs) in π-conjugated organic materials serve as tunable levers that influence molecular structure and intermolecular interactions in the condensed phase and, in turn, impact the electronic, optical and mechanical properties of these materials. NCIs include attractive dispersion, electrostatic and induction interactions as well as repulsive exchange interactions. 


