The increasing atmospheric CO2 concentration causes significant concern about global warming and its environmental impact. Industrial sectors, including power generation, transportation, and production factories, contribute most of the anthropogenic sources of CO2 emissions. Researchers search for methods to mitigate these CO2 emissions without compromising the benefits of industrial processes, including CO2 capture, utilization, and sequestration (CCUS). Post-combustion carbon capture (PCCC) by amine scrubbing is the most technology ready CCUS, with various pilot-scale projects worldwide showing small-scale to fully operational plants. While researchers have made significant progress in amine scrubbing, they strive to improve the efficiency and economics of these plants. One challenge they face includes solvent degradation due to flue gas constituents and temperature effects.
This dissertation focuses on the degradation byproducts' impact on the parent amine solvent. It explores knowledge about degradation byproducts in amine solvents used in carbon capture, including what degradation products, how much, how they form, and what impact these products have on the capture process. The dissertation splits into three sections where the first focuses on three thermal degradation byproducts of ethanolamine (MEA): oxazolidine-2-one (OZD), N-(2-hydroxyethyl)-ethylenediamine (HEEDA), and N-(2-hydroxyethyl)-imidazoline-2-one (HEIA) interactions in the solvent. The second focuses on O2 solubility and the role O2 plays in oxidative degradation. The final section discusses some initial degradation results of a new class of amine carbon capture solvents called water-lean (WL) solvents.
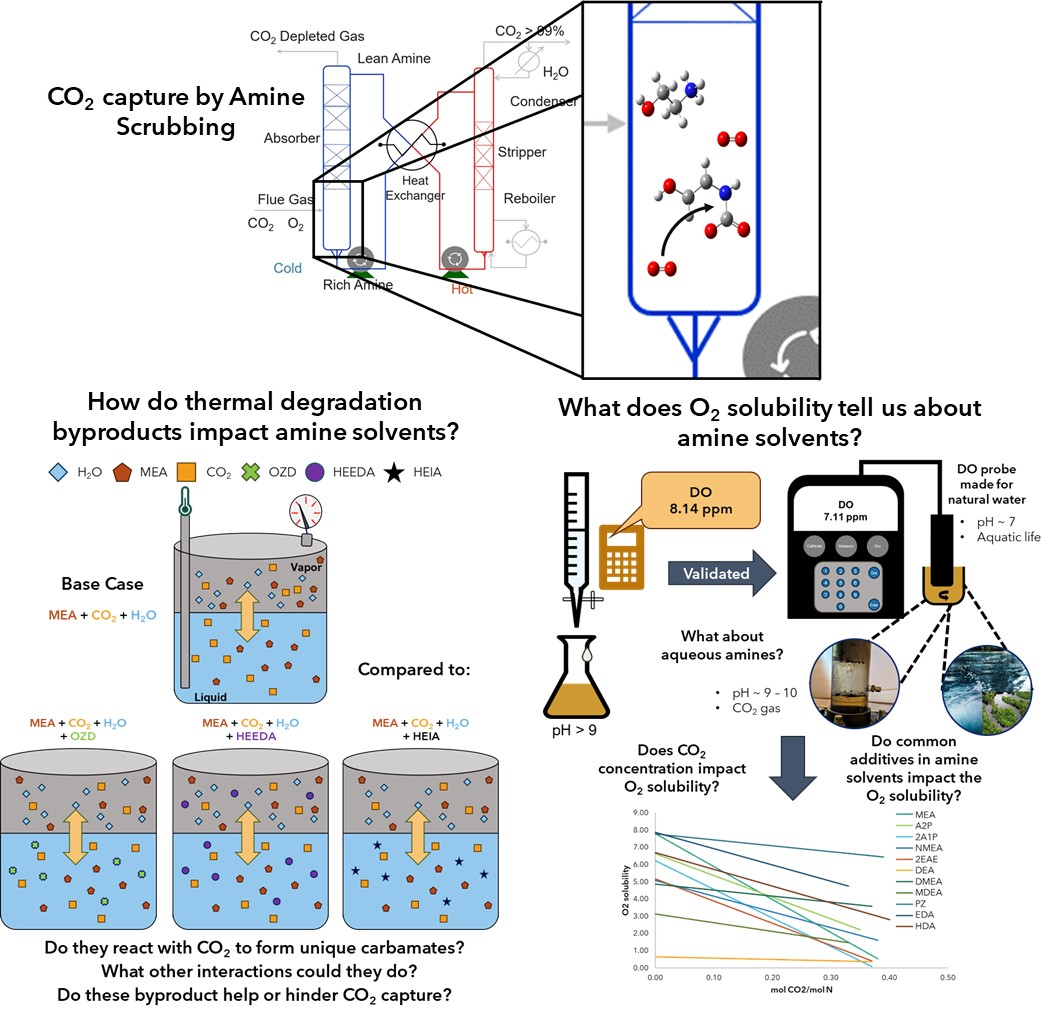
The discussion of the thermal degradation byproducts began by describing thermal degradation byproducts that react with CO2 with and without the parent amine present. The species found were explored further at the liquid-vapor barrier to explain how the species interact with each other. The degradation byproducts with unhindered primary or secondary amines, such as HEEDA, create a pseudo-blended solvent that could improve the solvent's capture performance as it improves the CO2 solubility in the amine solvents. In contrast, hindered structures like HEIA and OZD will minimally impact the speciation in the amine solvents. However, these species revert to the carbamate at lower CO2 concentrations or higher temperatures and further react with CO2 concentrations.
The discussion on O2 solubility in the different amine solvents and the role O2 plays in oxidative degradation began by validating a dissolved oxygen (DO) electrochemical probe by redox titrations following the Winkler method. The validated probe then measured the DO or O2 solubility in different amine solutions under various conditions found in CO2 capture. It also explored how additives used in advanced amine solvents impact the O2 solubility in the solution. The redox titration measured the DO slightly higher than the probe, validating the electrochemical probe for amine solvents. However, solvents with transition metals that can oxidize iodine cannot use this method to measure the DO in the solvents. In the amine solvents, the diamines measured higher O2 solubility than the alkanolamines, and the alkanolamines measured higher O2 solubility than the amine diols. In addition, as the number of hydrogens bonded to nitrogen decreased, the O2 solubility decreased. The ionic effect on O2 was also demonstrated, where the DO decreased with increased CO2 loading. However, there was still DO, indicating that the solvents will undergo oxidative degradation at high loadings. Furthermore, the solvents minimally impacted the O2 solubility except those that can react with CO2. The additive 2-mercaptobenzathiazole (MBT) decreased the DO concentration until it exhausted its capacity to react with O2, and then the DO rose back to the levels at equilibrium. The additive sodium metavanadate increased the DO concentration because metavanadate can react with O2, increasing the oxidative potential in the solution. The final section described the initial findings of degradation byproducts of WL solvents and what the organic cosolvent does in the system, including some initial discussions on how the organic cosolvent impacts an amine solvent, how the solvent phase separates at higher loadings due to an ionic effect in the cosolvent, and what compounds were found during oxidative degradation experiments.
KEYWORDS: Carbon Capture, Amine Scrubbing, Degradation, Amine Oxidative Degradation, Amine Thermal Degradation, Water Lean Solvents
