The question of how life has begun has fascinated scientists from many disciplines and it was the organic chemist Graham Cairns-Smith who proposed the theory for the origin of life starting from clays instead of polymers such as RNA.
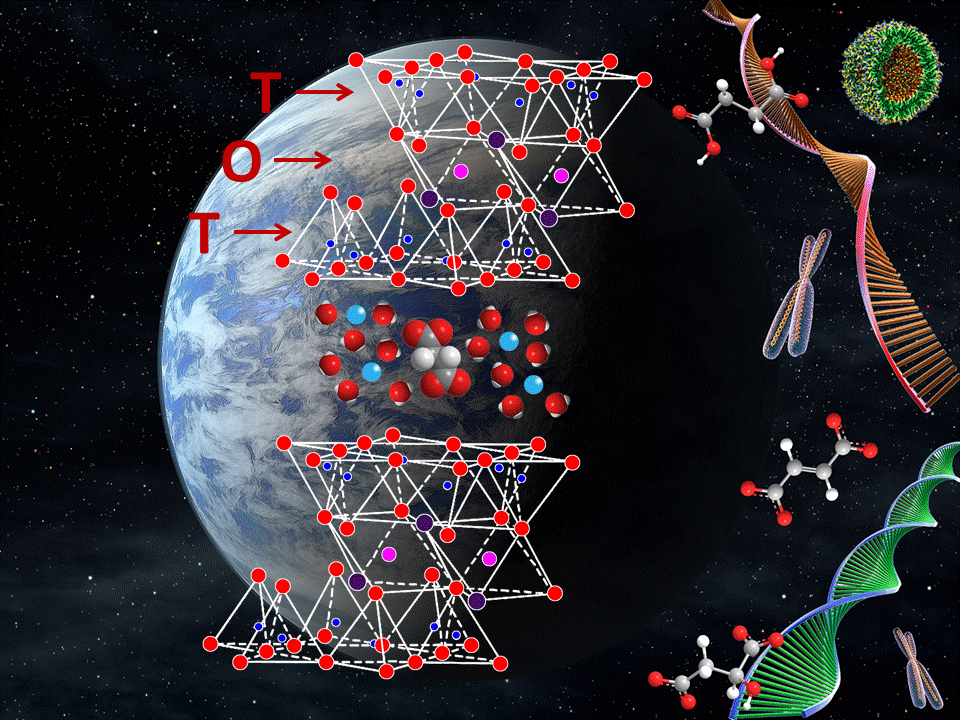
The source of the monomers such as nucleotides, amino acids and dicarboxylic acids were relegated by Cairns-Smith to the evolution of metabolism, which is the synthesis of amino acids and nucleotides from the citric acid cycle.
This problem of the evolution of metabolism has recently been advanced by the behavior of simple semiconductor minerals such as zinc sulfide (ZnS), which are capable of harvesting sunlight energy and converting this energy into the formation of chemical bonds of dicarboxylic acids from CO2 thus providing the core reactions of universal metabolism before the existence of enzymes.
A connection between ZnS prebiotic photosynthesis and clay replication has now been established in a paper published by a team of scientists from the University of Kentucky and the Massachusetts Institute of Technology (MIT) in the United Sates, and McGill University in Canada. The paper has related how prebiotic metabolites available from simple sunlight promoted reactions can catalyze the synthesis of clay minerals (i.e., a zinc clay called sauconite). The work shows that central metabolites such as succinate and malate can enable the nucleation process for clay formation. These prebiotic metabolites have been generated by photocatalysis with ZnS, and this work demonstrates how they can catalyze the synthesis of clays. The study published in the open access journal Scientific Reports shows how a clay synthesis can proceed catalyzed by prebiotic metabolites in only 20 hours at 90 °C and 1 atm. Clay formation generally requires much longer times as well as higher temperature and pressure. The cryogenic transmission electron microscopy clearly shows that clay nanoparticles can be observed after only 6 hours of synthesis, as verified by the incorporation of aluminum into the tetrahedral layer.
The team noted that the synthesis of clay can proceed at even lower temperatures, i.e., at just 70 °C, with the addition of a single seed particle. The work presents an excellent example of the reproductive power of clay minerals and the mechanism by which prebiotic metabolites catalyze their formation. Clay minerals acting as chemical sponges can retain water and polar organic molecules, and should have played a key role in the origin of life; 1) protecting against ultraviolet radiation, and 2) concentrating and catalyzing the polymerization of organic molecules such as RNA. The outcome of this work has direct implications to understand the origin of life on the early Earth and other rocky planets.
This research was supported in part by the U.S. National Science Foundation. Any opinions, findings, and conclusions or recommendations expressed in this article do not necessarily reflect the views of the National Science Foundation.
More information: Ruixin Zhou, Kaustuv Basu, Hyman Hartman, Christopher J. Matocha, S. Kelly Sears, Hojatollah Vali & Marcelo I. Guzman. Catalyzed Synthesis of Zinc Clays by Prebiotic Central Metabolites, Scientific Reports, 7, 533, 2017. DOI: 10.1038/s41598-017-00558-1.
Journal reference: Scientific Reports
This article is reproduced from the original published at phys.org
